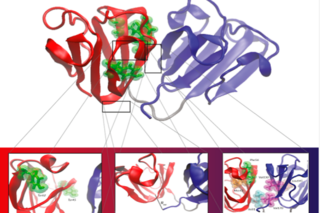
Human γD-crystallin structure highlighting the N-terminal domain aromatic residues and the hydrophobic residues of the interdomain interface.
The second law of thermodynamics predicts increasing disorder, yet living systems rely on highly organized structures ranging from molecular complexes to entire tissues. Although cells require energy to maintain this organization, many assembly processes can occur spontaneously through thermodynamic driving forces such as intermolecular interactions and concentration changes. These processes underlie essential phenomena like phase separation, membrane organization, and tissue formation, but can also lead to harmful outcomes such as protein aggregation and denaturation, which are linked to diseases including Alzheimer’s, Parkinson’s, ALS, and cataracts.
Our group uses computational methods based on molecular thermodynamics and statistical physics to study the kinetics of biomolecular aggregation. We focus in particular on γ-crystallin, a lens protein associated with age-related cataracts, to understand the timescales and mechanisms of rare aggregation events. This work aims to provide insights that may support early disease detection and the development of therapeutic strategies.
Related Work
-
36. Effect of Pressure on the Conformational Landscape of Human γD-Crystallin
Kacirani A., Uralcan B., Domingues T. D., Haji-Akbari A. J. Phys. Chem. B (2024) 128, 20, 4931–4942.
-
22. Thermodynamically Driven Assemblies and Liquid-liquid Phase Separations in Biology.
Falahati H, Haji-Akbari, A. Soft Matter 15: 1135 (2019)